12.04.2024
Efficient Tandem Cu-Catalyzed Click Synthesis of Multi-Sugar-Modified Oligonucleotides.
Angewandte Chemie Internation Edition
Tölke AJ, Gaisbauer JF, Gärtner YV, Steigenberger B, Holovan A, Streshnev F, Schneider S, Müller M, Carell T.
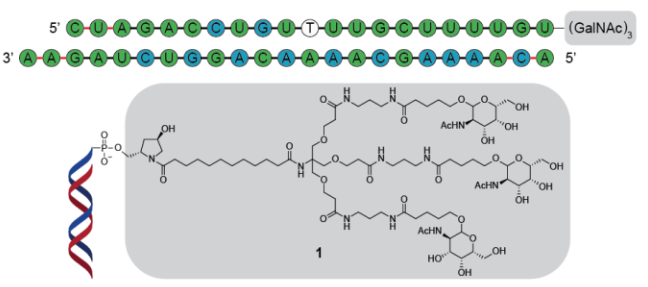
Nucleic acids in the form of siRNA, antisense oligonucleotides or mRNA are currently explored as new promising modalities in the pharmaceutical industry. Particularly, the success of mRNA-vaccines against SARS-CoV-2, along with the successful development of the first sugar-modified siRNA therapeutics has inspired the field. The development of nucleic acid therapeutics requires efficient chemistry to link oligonucleotides to chemical structures that can improve stability, boost cellular uptake, or enable specific targeting. For the siRNA therapeutics currently in use, modification of the 3′-end of the oligonucleotides with triple-N-acetylgalactosamine (GalNAc)3 was shown to be of significance. This modification is currently achieved via a cumbersome multi-step synthesis and subsequent loading onto the solid support material. Here, we report the development of a bifunctional click-reactive linker that allows the modification of oligonucleotides in a tandem click reaction with multiple sugars, regardless of the position within the oligonucleotide, with remarkable efficiency and in a one-pot reaction.

Proof of Concept Grant for Thomas Carell
The European Research Council has awarded the LMU chemist a grant for the preclinical development of...

LMU Spin-off RNhale gegründet
The formation of the biotech spinoff RNhale can be traced back to Professor Olivia Merkel's groundbreaking...

EpiCure gewinnt 2. Runde beim Businessplanwettbewerb
EpiCure, eine Firmenidee, die auf Ausgründung wartet, gehört mir ihrem Konzept zu den 5 Siegern...

EisbachBio GmbH: Allosterischen Inhibitor erhält FDA-Freigabe
Eisbach Bio GmbH (“Eisbach” or the “Company”), a privately-held clinical-stage biotechnology company...

LMU Spin-off: Gründung der QuGen GmbH
Die QuGen GmbH, gegründet von Thomas Carell und Pascal Giehr, bietet hoch-sensitive Long-read Sequenzierung...
