Nucleic acid-based agents for difficult-to-treat diseases
Infections and inflammations as well as tumor diseases and diseases of the cardiovascular system together account for about 75% of all deaths in the EU. Secondary diseases, such as heart failure in Duchenne muscular dystrophy, also contribute significantly to mortality.
Available therapies in these disease areas are often not sufficiently efficient or are limited to symptom relief, without having a lasting effect on the progression of the disease. Therefore there is a considerable medical need in these areas.
In CNATM, we are moving away from traditional protein-centric drug discovery and aim to develop nucleic acids as therapeutics. Nucleic acids are the most promising class of molecules to achieve rapid development of highly specific therapeutics - as already demonstrated by the mRNA-based COVID-19 vaccines. In CNATM, we exploit an even broader spectrum, including chemically modified mono- and dinucleotides, oligonucleotides such as siRNAs or antimire, and aptamers.
With currently 11 projects, we are pursuing the ambitious goal of moving 10 active substances from development to validation and further to preclinical testing within a few years.
In all projects very promising preliminary results have been achieved. Some of the projects are already so far advanced that we will accelerate them into validation as "fast track projects". Our 16 industrial partners are closely involved from the outset and will make a significant contribution with research activities such as, syntheses and active ingredient formulations.
Mono- and dinucleotides
Decitabine is a synthetic nucleoside that can significantly slow the progression of acute myeloid leukaemia (AML) through an epigenetic mechanism that is not fully understood. However, this is not seen in all patients and toxic side effects are common. Preliminary work in WP1 has shown that decitabine can be chemically modified to increase its stability and reduce unwanted side effects. The aim of this WP is to develop decitabine analogs with even better pharmacological properties.
Head: Thomas Carell

PIs: Thomas Carell, Pascal Giehr, Irmela Jeremias, Pavel Kielowski, Franziska Traube
Partners:

Stimulator of Interferon Genes (STING) is a receptor that plays a significant role in inflammatory and degenerative diseases. Its natural ligand is cyclic guanosine adenosine monophosphate (cGAMP), which is produced by cGAMP synthase in response to DNA mutations. The goal of this WP is to develop synthetic cGAMP analogs that act antagonistically on STING and are suitable for oral therapy of AML.
Head:
Karl-Peter Hopfner

Veit Hornung

PIs: Karl-Peter Hopfner, Veit Hornung, Thomas Carell, Irmela Jeremias, Heinrich Leonhardt, Marion Subklewe
Partners:


Oligonucleotides
Chronic obstructive pulmonary disease (COPD) is the third leading cause of death worldwide. Currently, there is no therapy that can interfere with the underlying inflammatory processes of COPD. Preliminary work on WP6 showed that the alveolar macrophage and its non-coding RNAs (ncRNAs) have a central role in the development of COPD. The aim of this WP is to develop antisense oligonucleotides against ncRNAs that, by coupling to a ligand, achieve enhanced uptake in alveolar macrophages and thus locally increased drug levels.
Head: Stefan Engelhardt

PIs: Stefan Engelhardt, Ali Önder Yildirim
Partners:


WP7 focuses on the observation that macrophages may have a significant role in the development of certain tumors, thus linking them to inflammatory processes. There is increasing evidence that long non-coding RNAs (lncRNAs) control essential functions of macrophages and thus have therapeutic potential. Building on a CRISPR-Cas-based screening of thousands of lncRNAs established by AG Rad, suitable targets will be identified and eventually antisense oligonucleotides against these lncRNAs will be developed.
Head: Roland Rad

PIs: Roland Rad, Stefan Engelhardt, Annalisa Marsico, Gunther Meister, Michael Sattler
Partners:

The widespread use of classical antibiotics carries the risk of multi-resistance and also leads to unwanted side effects such as damage to the natural bacterial flora. In this subproject, highly selective RNA antibiotics are being developed as a novel approach for the targeted control of pathogenic bacteria. Antisense oligonucleotides are coupled to peptides that bind to specific surface structures of the bacteria, thus enabling the oligonucleotide to be introduced.
Head: Jörg Vogel

Delivery and formulation of nucleic acid therapeutics
While mRNA vaccines were important in the containment of the COVID-19 pandemic, no antivirals with sufficient efficacy are available against SARS-CoV2. The development of synthetic small interfering RNAs (siRNAs) against virally encoded RNAs has shown initial success, but currently no clinically approved siRNAs with antiviral efficacy are available. WP3 therefore aims to develop chemically modified siRNA conjugates and siRNA formulations with enhanced target specificity. We expect that the results of this WP will result in new approaches for siRNA-based therapy of other viral infections.
Head: Olivia Merkel

PIs: Olivia Merkel, Thomas Carell, Olivia Keppler, Sabine Schneider, Philip Tinnefeld
Partners:



One of the key challenges for nucleic acid therapeutics is their delivery into cells. The embedding of nucleic acids in liposomal nanoparticles is often chosen for this purpose - a proven approach, which, however, still leaves open wishes with regard to uptake efficiency and cell/tissue specificity. WP4 applies defined test systems in which molecular libraries of carrier sequences or chemical modifications are iteratively investigated and developed for different applications (siRNA, mRNA, different organs and application routes).
Head: Ernst Wagner

PIs: Ernst Wagner, Olivia Merkel
Partners:


mRNA agents and other principles
The replacement of uridine by 1-methylpseudouridine helped mRNA vaccines against COVID-19 to achieve significantly improved pharmacological properties. However, the limited shelf life and laborious formulation in liposomes complicate the production and presumably on the efficiency of mRNA vaccines. It can be expected that further modifications will allow considerable optimizations. The focus of this project is the advancement and use of chemical modification of nucleotide positions of the mRNA (based on click chemistry) as well as the generation of polycistronic mRNAs in order to be able to vaccinate against co-existing virus variants with the aid of a single vaccine.
Head: Thomas Carell

PIs: Thomas Carell, Veit Hornung, Pavel Kielkowski
Partner:


WP8 addresses two significant and often fatal cardiovascular diseases: Carotid artery stenoses and aneurysms of the abdominal aorta. In both cases, changes in vascular structure are causative for the disease. Preliminary work on WP8 already showed that a small non-coding RNA, the microRNA miR-126-5p, in the cell nucleus can assume an unusual function as an RNA aptamer that has a protective effect on endothelial cells. Here, the binding of the microRNA to the protein Mex3A is crucial. The aim of this WP is to develop aptamers that preferentially bind to Mex3A and, based on this, to develop drug-eluting stents that release the aptamer as a protective agent.
Head: Lars Mägdefessel

WP9 is dedicated to cardiovascular diseases, in this case in the context of skeletal muscle disease. Specific guide RNAs for CRISPR/Cas-based gene therapy of DMD will be developed and formulated into nanoparticles. Initially, the therapy will be studied experimentally in organoid models before final validation in large animal models. Since the dystrophin gene responsible for DMD is located on the X chromosome, WP9 also aims to activate the intact dystrophin allele on the second (inactive) X chromosome by manipulating epigenetic factors.
Head: Alessandra Moretti

PIs: Alessandra Moretti, Daniel Andergassen, Christian Kupatt
Partners:
Hepatitis B is a very common chronic viral disease that is difficult to cure. The established therapy is based on the use of nucleoside analogous that can control virus replication but are not curative. Interferon alpha may cure hepatitis B, but is accompanied by high side effects as it is applied systemically. The aim of this sub-project is to package interferon-encoding mRNAs into nanoparticles and deliver them directly to HBV-infected livers.
Head: Arne Skerra

PIs: Arne Skerra, Ulrike Protzer, Carina de Oliveira Mann
Partners:


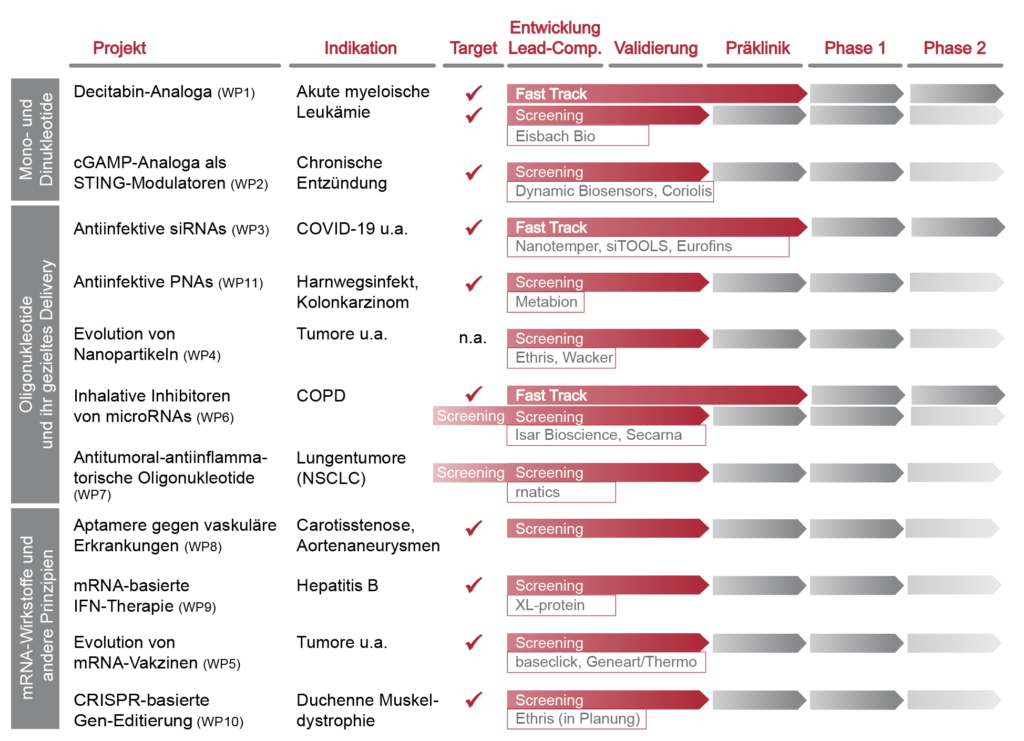
Project overview and pipeline of drug development in the CNATM Future Cluster:
Red time arrows describe activities of the 1st implementation phase, gray those of the subsequent ones.
Company collaborations during the 1st implementation phase are assigned to the WP's.
VP: Collaborative project as organizational assignment
